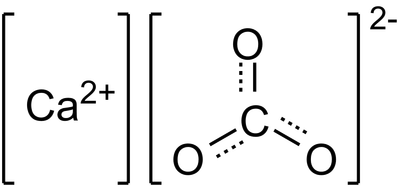
The chemical name for Tums is calcium carbonate. There are some common names such as Tums, Chalk e.g. The chemical formula for Calcium carbonate is CaCO3. The elements that make the compound CaCO3 are calcium, carbon, and oxygen.
Physical Properties
The chemical properties of Tums or calcium carbonate is that it is insoluble in water. Some physical properties is that it is solid, and odourless. When it reaches boiling point it starts to decompose and its melting point is 1517-2442°F. It is a white hexagonal crystal or powder. The taste of calcium carbonate is that it has a chalk taste but others taste it as tasteless.
Chemical Properties
The chemical properties of Tums or calcium carbonate is that it is insoluble in water. The exact mass of Calcium Carbonate is 99.947335 g/mol and the molecular weight is 100.0869 g/mol. The density is 2.7-2.95cm3. When calcium carbonate is heated to decomposition it starts to emit acrid smoke and irritating vapors. Calcium carbonate is not corrosive.
Physical Properties
The chemical properties of Tums or calcium carbonate is that it is insoluble in water. Some physical properties is that it is solid, and odourless. When it reaches boiling point it starts to decompose and its melting point is 1517-2442°F. It is a white hexagonal crystal or powder. The taste of calcium carbonate is that it has a chalk taste but others taste it as tasteless.
Chemical Properties
The chemical properties of Tums or calcium carbonate is that it is insoluble in water. The exact mass of Calcium Carbonate is 99.947335 g/mol and the molecular weight is 100.0869 g/mol. The density is 2.7-2.95cm3. When calcium carbonate is heated to decomposition it starts to emit acrid smoke and irritating vapors. Calcium carbonate is not corrosive.